提取条件和提取物的处理:通过浸泡或灌装一次性组件(袋或管)来提取一次性组件。刚性一次性组件,如过滤器和外壳,通过摇动彻底湿润,以降低一次性组件和溶剂之间的界面阻力,并使表面易于接触溶剂。只要有可能达到所需的SA/V比,一次性组件就可无须分割整个使用。不执行切碎等操作。按照预期用途对组件进行处理:对于使用前可能经过辐照和高压灭菌的组件,提供每个预处理步骤的数据。按照说明书冲洗用于保存一次性组件的液体(如切向流盒、膜吸附器)。使用已清洁的设备进行提取。空白样品、样品制备和测量细节见Menzel等人的文章(5)。关于根据实验室工作的基本原则处理提取物的其他建议可在文献中找到
(17, 18, 21)。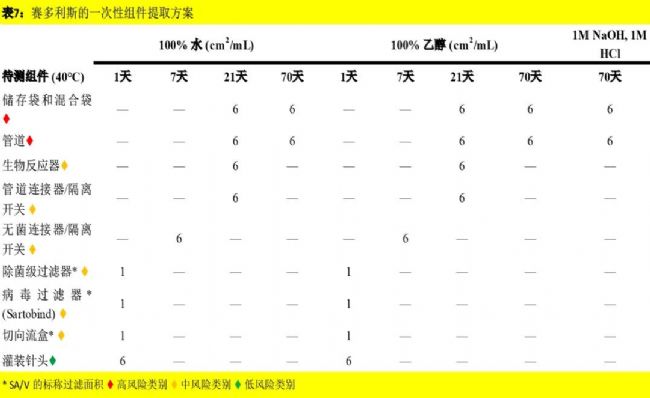
4. 分析方法
我们结合了最先进的分析技术,用于检测、鉴定和定量挥发性、半挥发性和非挥发性可提取物,包括元素。我们的分析方法如表5所示。
报告限的定义:美国药典第<1663> 章提到“表征是发现、鉴定和量化超过规定水平或阈值的提取物中存在的每个有机和无机化学实体。这些阈值可以基于患者安全考虑、材料考虑、分析技术能力等”(16)。许多文献描述了用不同分析方法测定可提取化合物的检出限(LoD)和定量限(LoQ)的适用方法(22, 23)。Jenke等人报道了一次性组件中约500种不同的潜在可提取化合物(24)。由于所列可提取化合物的极性和挥发性的化学多样性,不能期望LoD/LoQ值在相同或甚至相似的水平上。美国药典第<1663>章讨论了定性可提取物评估,并建议至少有一种浓度为5µg/mL的可提取化合物来进行结构确证。
在可提取物研究中,扫描方法允许检测浓度范围为十亿分之几(ppb)到百万分之几(ppm)的潜在可提取化合物。为了能够稳健地报告可提取物结果(包括定性和定量),定义每种分析方法的报告限(RL)是一个实用步骤。这些限值是主观定义的,对于单一化合物可以高于定量限,并且可以克服实验室间定量限的差异。RL可以从特定分析技术的单个化合物的LoQ数据中得到。这一概念允许报告来自不同实验室的可重复的可提取物信息。
在研究中,从提取样品中检测到的所有峰,如果峰面积超过对照峰(空白)峰面积的50%,则视为可提取化合物。RL不是固定的,代表分析设备的性能(表6)。进一步的改进和新的耐用的分析系统和技术可以导致较低的报告限。
赛多利斯的一次性组件提取方案:表7显示了应用于一次性组件的提取方案。赛多利斯在其标准、可配置和自定义一次性组装中使用了许多第三方组件,包括连接器和管道。为了向我们的客户提供我们的一次性系统的全面可提取物信息,我们实施了一个全面的计划,根据我们新的内部程序测试我们组件库的一个子集(包括此类第三方组件)。
赛多利斯已经开发出一种可提取物研究的实用方法,以表征用于生物制药工艺的一次性组件的潜在可提取物。同时建立了一个测试程序,以评估提取过程中物理和化学参数的影响,并推导出不同一次性组件提取物研究设计的相关条件。通过采用标准化提取参数和最先进的分析方法对一次性组件进行的最差情况提取研究的结果,赛多利斯能够帮助您获得全面的定性和定量可提取物数据。
(点击获得完整验证服务)
查询原文
Pahl I., Dorey S., Uettwiller I., Hoffmann Ch., Priebe P., Menzel R.,
& Hauk A.; Development of a Standardized Extractables Approach for
Single-Use Components -General Considerations and Practical Aspects.
Bioprocess Int. 2018;16(10).
以上作者均来自赛多利斯
参考文献
1.Reif OW, Sölkner P, Rupp J. Analysis and Evaluation of Filter
Cartridge Extractables for Validation in Pharmaceutical Downstream
Processing. PDA J. Pharm. Sci. Technol. 50(6) 1996; 399–410.
2.Fichtner S, et al. Determination of “Extractables” on Polymer
Materials by Means of HPLC-MS. PDA J. Pharm. Sci. Technol. 60, 2006;
291–301.
3.Pahl I, et al. Analysis and Evaluation of Single-Use Bag Extractables
for Validation in Biopharmaceutical Applications. PDA J. Pharm. Sci.
Technol. 68(5) 2014: 456–471; doi:10.5731/ pdajpst.2014.00996.
4.Menzel R, et al. Comparative Extractables Study of Autoclavable
Polyethersulfone Filter Cartridges for Sterile Filtration. PDA J. Pharm.
Sci. Technol. 72(3) 2018: 298–316; doi:10.5731/pdajpst.2017.008367.
5.Dorey S, et al. Theoretical and Practical Considerations When
Selecting Solvents for Use in Extractables Studies of Polymeric Contact
Materials in Single-Use Systems Applied in the Production of
Biopharmaceuticals. Ind. Eng. Chem. Res. 57, 2018; 7077–7089;
doi:10.1021/acs.iecr.7b04940.
6.Hauk A, et al. On the “Fate of Leachables” in Biopharmaceutical
Up-Stream and Down-Stream Processes. Single-Use Technologies II:
Bridging Polymer Science to Biotechnology Applications. ECI Conference
Series: 7–10 May 2017, Tomar, Portugal.
7.Gaston F, et al. FTIR Study of Ageing of γ-Irradiated
Biopharmaceutical EVA Based Film. Polym. Degrad. Stab. 129, 2016; 19–25;
doi:10.1016/j.polymdegradstab.2016.03.040.
8.Audran G, et al. Degradation of γ-Irradiated Polyethylene-Ethylene
Vinyl Alcohol-Polyethylene Multilayer Films: An ESR Study. Polym.
Degrad. Stab. 122, 2015; 169– 179;
doi:10.1016/j.polymdegradstab.2015.10.021.
9.Gaston F, et al. Impact of γ-Irradiation, Ageing and Their
Interactions on Multilayer Films Followed By AComDim. Anal. Chim. Acta
981, June 2017: 11–23; doi:10.1016/j.aca.2017.05.021.
10.Gaston F, et al. One Year Monitoring By FTIR of γ-Irradiated
Multilayer Film PE/EVOH/PE. Radiat. Phys. Chem. 125, 2016: 115–121;
doi:10.1016/j. radphyschem.2016.03.010.
11.Merseburger T, et al. A Risk Analysis for Production Processes with
Disposable Bioreactors. Disposable Bioreactors 2. Eibl D, Eibl R, Eds.
Springer: Berlin–Heidelberg, 2013: 273– 288; doi:10.1007/10_2013_244.
12.Merseburger T, et al. Recommendation for a Risk Analysis for
Production Processes with Disposable Bioreactors. DECHEMA, Gesellschaft
für Chemische Technik und Biotechnologie eV: Frankfurt am Main, Germany,
2015.
13.<661> Plastic Packaging Systems and Their Materials of Construction. United States Pharmacopeia 40(1) 2017.
14.<665> DRAFT. Polymeric Components and Systems Used in the
Manufacturing of Pharmaceutical and Biopharmaceutical Drug Products. US
Pharmacopeial Convention, Inc.: Rockville, MD, 2017
15.Plastic Packaging: Interactions with Food and Pharmaceuticals.
Piringer OG, Barner AL, Eds. Wiley‐VCH: Weinheim, Germany, 2008.
16. <1663> Assessment of Extractables Associated with
Pharmaceutical Packaging/Delivery Systems. United States Pharmacopeia
38, 2015: 7166–7180.
17.Leachables and Extractables Handbook: Safety Evaluation,
Qualification, and Best Practices Applied to Inhalation Drug Products.
Ball DJ, et al., Eds. John Wiley & Sons, Inc.: Hoboken, NJ, 2012.
18.Jenke D. Compatibility of Pharmaceutical Products and Contact
Materials: Safety Considerations Associated with Extractables and
Leachables. John Wiley & Sons, Inc.: Hoboken, NJ, 2009.
19.Dorey S, et al. Reconciliation of pH, Conductivity, Total Organic
Carbon with Carboxylic Acids Detected By Ion Chromatography in Solution
After Contact with Multilayer Films After γ-Irradiation. Eur. J. Pharm.
Sci. 117, 23 February 2018; 216–226; doi:10.1016/j.ejps.2018.02.023.
20.ISO 11137-1:2006. Sterilization of Health Care Products — Radiation —
Part 1: Requirements for Development, Validation, and Routine Control
of a Sterilization Process for Medical Devices. International
Organization for Standardization: Geneva, Switzerland, 2016.
21.Jenke D, et al. Extractables Characterization for Five Materials of
Construction Representative of Packaging Systems Used for Parenteral and
Ophthalmic Drug Products. PDA J. Pharm. Sci. Technol. 67(5) 2013;
448–511; doi:10.5731/ pdajpst.2013.00933.
22.Shrivastava A, Gupta V. Methods for the Determination of Limit of
Detection and Limit of Quantitation of the Analytical Methods.
Chronicles Young Sci. 2(1) 2011; 21–25; doi:10.4103/2229-5186.79345.
23.ICH Q2(R1). Validation of Analytical Procedures: Text and
Methodology. US Fed. Reg. 62(96) 1997: 27463–27467;
www.ich.org/fileadmin/Public_Web_Site/ICH_Products/
Guidelines/Quality/Q2_R1/Step4/Q2_R1__Guideline.pdf.
24.Jenke D, Carlson T. A Compilation of Safety Impact Information for
Extractables Associated with Materials Used in Pharmaceutical Packaging,
Delivery, Administration, and Manufacturing Systems. J. Pharm. Sci.
Technol. 68(5) 2014: 407–55; doi:10.5731/pdajpst.2014.00995.