Flow cytometry analysis of the quantitative expression of 3 key pluripotency-associated transcription factors (Nanog, OCT3/4 and SOX2) complemented the image-based analysis (Figure 5). Even after long-term expansion on the FN1 motifs surface, hiPSCs still exhibited a high level of Nanog, OCT3/4and SOX2 expression (> 95% positive cells in the entire population). The pluripotency marker expression profle was similar to that observed in hiPSCs expanded on the Corning Matrigel-coated surface.

Figure 5: Flow cytometry analysis of pluripotency markers after long-term expansion of hiPSCs
After 24 successive passages on the Eppendorf CCCadvanced FN1 motifs
surface, > 95% of the total hiPSC population expressed the
pluripotency markers Nanog, OCT3/4 and SOX2, as evaluated by flow
cytometry analysis. This pluripotency marker expression profle was
comparable to hiPSCs cultured on a biological Corning Matrigel-coated
surface. Isotype controls validated the specifcity of the stain.
Unstained cells (indicated in red) determined the percentage of positive
cells for the three markers of interest (indicated in black).
The functional pluripotency of hiPSCs after long-term expansion on the
FN1 motifs surface was evaluated by their in vitro differentiation into
cells of the three embryonic germ layers: endoderm, mesoderm and
ectoderm (Figure 6). As suggested by positive specifc staining of
well-established markers of the three embryonic germ layers, hiPSCs
preserved their trilineage differentiation potential even after 20
successive passages on the FN1 motifs surface.
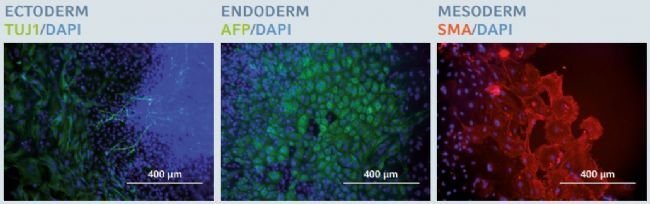
Figure 6: Trilineage differentiation potential after long-term expansion of hiPSCs
After 20 successive passages on the Eppendorf CCCadvanced FN1 motifs
surface, hiPSCs maintained their trilineage differentiation potential, as
examined by specifc fluorescent staining of three specifc embryonic germ
layer markers: β-III tubulin (TUJ1) in ectoderm, alpha-fetoprotein
(AFP) in endoderm and smooth muscle actin (SMA) in mesoderm. Cells were
counterstained with a classic nuclear marker, DAPI. Scale bar indicates
400 µm.
Genomic stability after long-term xeno-free expansion on the Eppendorf CCCadvanced™ FN1 motifs surface
Long-term expansion of PSCs under feeder-free conditions can be
responsible for the occurrence of chromosomal ab normalities [10]. In
order to monitor the genomic integrity of hiPSCs after long-term
expansion on the FN1 motifs surface in xeno-free culture medium, a
G-banding karyotype analysis was performed on fxed metaphase-blocked
cell samples obtained after 20 successive passages. Expanded hiPSCs
maintained a normal human karyotype (46XY) without chromosomal
abnormalities, thus confrming the genomic stability of hiPSCs after
long-term expansion on the FN1 motifs surface (Figure 7).
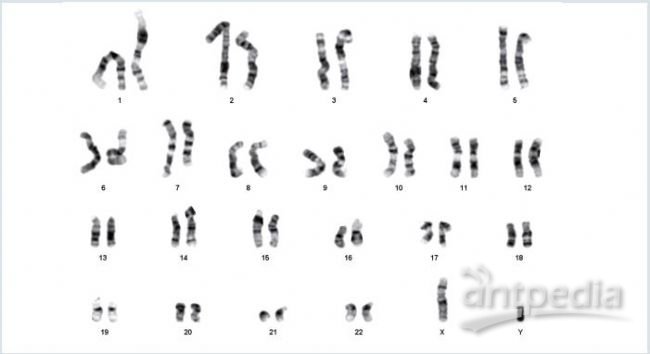
Figure 7: Karyotype analysis after long-term expansion of hiPSCs
After 20 successive passages on the Eppendorf CCCadvanced FN1 motifs
surface, hiPSCs exhibited a normal G-banding karyotype without any
chromosomal aberrations (46XY).
Conclusion
The ready-to-use Eppendorf CCCadvanced FN1 motifs surface supports
efcient, long-term hiPSC expansion in a completely defned, animal- and
human-component-free culture system. Its use is associated with a
consistent and robust growth rate of hiPSCs displaying their
characteristic morphology.
During the expansion process across at least 20 successive passages on
the FN1 motifs surface, hiPSCs remain undiffer entiated and retain all
pluripotent stem cell-specifc features, including the trilineage
differentiation potential, as well as their genomic integrity.
Literature
[1] Kumar D, Anand T, Kues WA. Clinical potential of human-induced
pluripotent stem cells: Perspectives of induced pluripotent stem cells.
Cell Biology and Toxicology 2017; 33(2): 99-112.
[2] Bilic J, Izpisua Belomonte JC. Concise review: induced pluripotent
stem cells versus embryonic stem cells: close enough or yet too far
apart? Stem cells 2012; 30(1): 33-41.
[3] Celiz AD, Smith JG, Langer R, Anderson DG, Winkler DA, Barrett DA,
Davies MC, Young LE, Denning C, Alexander MR. Materials for stem cell
factories of the future. Nature Materials 2014; 13(6): 570-579.
[4] Villa-Diaz LG, Pacut C, Slawny NA, Ding J, O’Shea KS, Smith GD.
Analysis of the factors that limit the ability of feeder cells to
maintain the undifferentiated state of human embryonic stem cells. Stem
Cells Development 2009; 18(4): 641-651.
[5] Xu C, Inokuma MS, Denham J, Golds K, Kundu P, Gold JD, Carpenter MK.
Feeder-free growth of undifferentiated human embryonic stem cells.
Nature Biotechnology 2001; 19(10): 971-974.
[6] Tong Z, Solanki A, Hamilos A, Levy O, Wen K, Yin X, Karp JM.
Application of biomaterials to advance induced pluripotent stem cell
research and therapy. EMBO 2015; 34(8): 987-1008.
[7] Villa-Diaz LG, Nandivada H, Ding J, Nogueira-de-Souza NC, Krebsbach
PH, O’Shea KS, Lahann J, Smith GD. Synthetic polymer coatings for
long-term growth of human embryonic stem cells. Nature Biotechnology
2010; 28(6): 581-583.
[8] Yang K, Lee J, Cho S-W. Engineering biomaterials for feeder-free
maintenance of human pluripotent stem cells. International Journal of
Stem Cells 2012; 5(1): 1-5.
[9] Lambshead JW, Meagher L, O’Brien C, Laslett AL. Defning synthetic
surfaces for human pluripotent stem cell culture. Cell Regeneration
2013; 2(1): 7.
[10] Rosler ES, Fisk J, Ares X, Irving J, Miura T, Rao MS, Carpenter MK.
Long-term culture of human embryonic stem cells in feeder-free
conditions. Developmental Dynamics 2004; 229(2): 259-274.